30 Nov 2017
Molecular diagnosis of spinal muscular atrophy with lower extremity predominance by NGS analysis
Authors:
Michela Catteruccia (michela.catteruccia@gmail.com), Michela Catteruccia, Fabiana Fattori, Adele D’Amico, Enrico Bertini
Unit of Neuromuscular and Neurodegenerative Disorders, Department of Neurosciences, Bambino Gesù Children’s Hospital, IRCCS, Rome, Italy
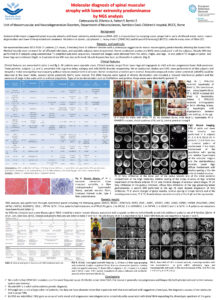
Background: spinal muscular atrophy with lower extremity predominance (SMA-LED) is characterized by congenital or early childhood onset motor neuron degeneration with lower limbs predominant weakness.
Methods: we examined patients with a phenotype suggestive for motor neuronopathy predominantly affecting the lower limbs were identified and referred for molecular diagnosis by Next-Generation Sequencing (NGS) panel including the following genes ASAH1; BICD2 ; DYNC1H1; EGR2; FIG4 ; GARS ; GDAP1; GJB1; HSN2; HSPB1; HSPB8; KIAA1985; LITAF; MFN2; PMP22; IGHMBP2; TBCE ; TRPV4.
Results: We report on 15 patients (11 males, 4 females) from 15 different families and characterized by congenital or childhood-onset lower limb wasting and weakness. All patients were sporadic cases. Clinical severity ranged from lower legs arthrogryposis to mild and non-progressive lower limb weakness. One patient had also epilepsy. Four patients underwent brain MRI 4/9 that revealed in one case a polymicrogyric pattern. ENG/EMG showed in all patients a neurogenic pattern of chronic denervation with reduced CMAP. Muscle biopsy was performed in 3/9 and showed neurogenic features. Seven patients underwent muscle MRI that showed in 5/6 a characteristic pattern of involvement of the thigh and the lower leg muscles. Molecular analysis showed that 3/15 had mutations in DYHC1H1, 2/15 a mutation in BICD2, 1/15 a mutation in SETX, 1/15 a mutation in TRPV4, 1/15 a mutation in GDAP1, 1/15 a mutation in FIG4, 2/15 were mutated in IGHMBP2. In 4/15 patients no mutations were identified.
By WES we characterized a new disease gene TBCE related to a motor neuron disease associated with a spastic ataxic syndrome of infantile onset in 5 patients out of 4 families.
Conclusion: Our findings further expands clinical and mutational spectra of SMA-LED. Although the cohort is not large, our data suggest that in well characterized patients, the diagnostic yield of our molecular test is over 70%.